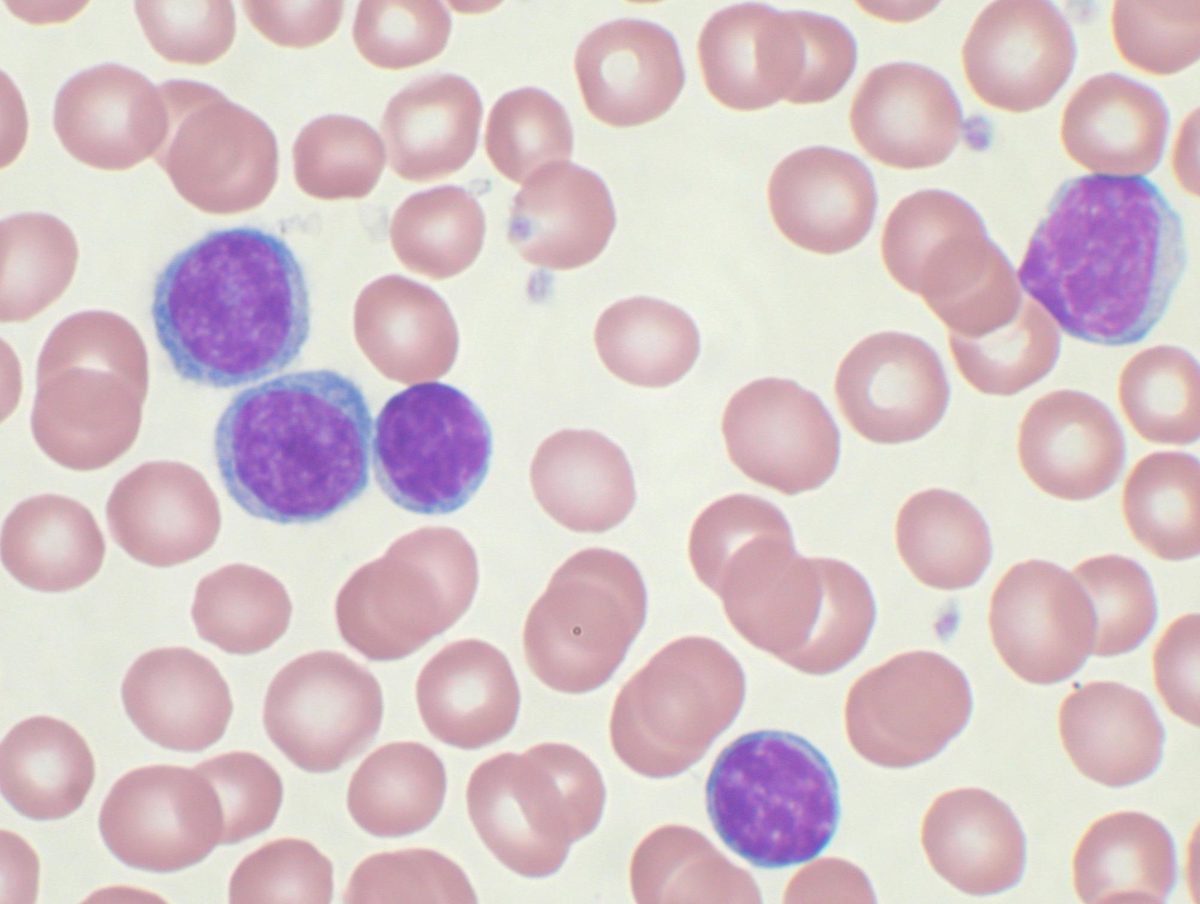
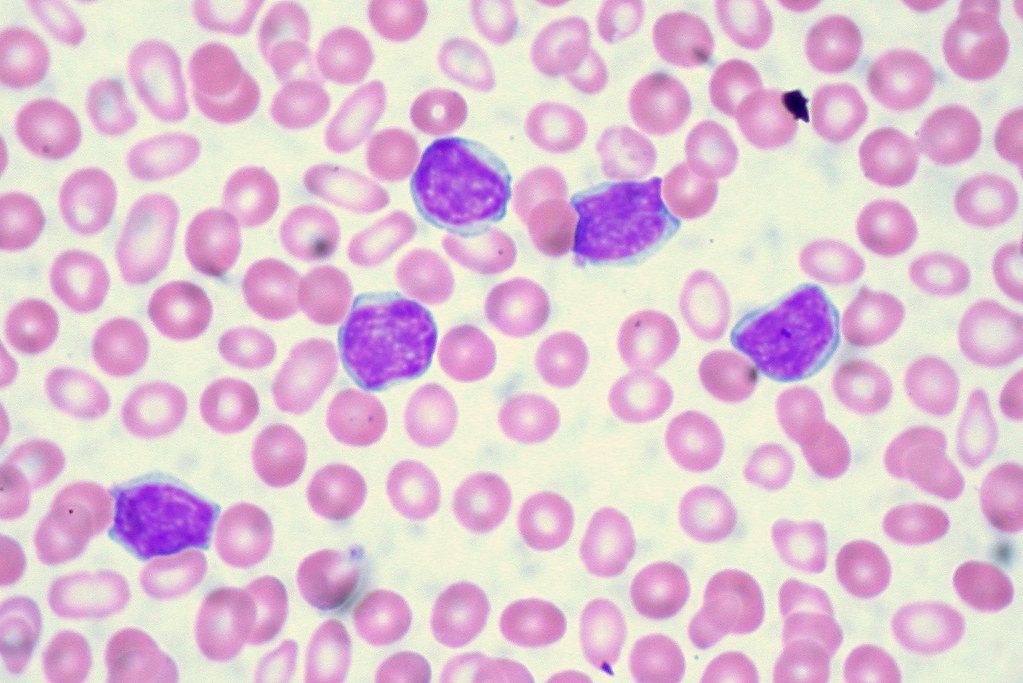
For decades, patients diagnosed with chronic lymphocytic leukemia—the most common adult leukemia in Western countries, affecting roughly 23,000 Americans each year—faced a difficult choice: endure rounds of intravenous chemotherapy with harsh side effects, or take targeted pills indefinitely, sometimes for life. On February 20, 2026, the Food and Drug Administration (FDA) approved a combination of two oral drugs, venetoclax and acalabrutinib, that eliminates both burdens. Patients take pills for roughly one year, then stop. In a trial of 867 patients, 77% remained cancer-free at three years.
The approval marks the first time an all-oral, fixed-duration regimen has been cleared for previously untreated chronic lymphocytic leukemia (CLL). It caps a twelve-year transformation in how this disease is treated—from toxic chemotherapy cocktails delivered through IV lines, to precisely targeted pills that block two specific proteins cancer cells need to survive. Each drug attacks a different survival mechanism: acalabrutinib disables a signaling enzyme called Bruton's tyrosine kinase (BTK) that tells cancer cells to multiply, while venetoclax blocks a protein called BCL-2 that prevents cancer cells from dying naturally. Together, they reduced the risk of disease progression or death by 35% compared to standard chemoimmunotherapy.