FDA approves camizestrant with safety labeling
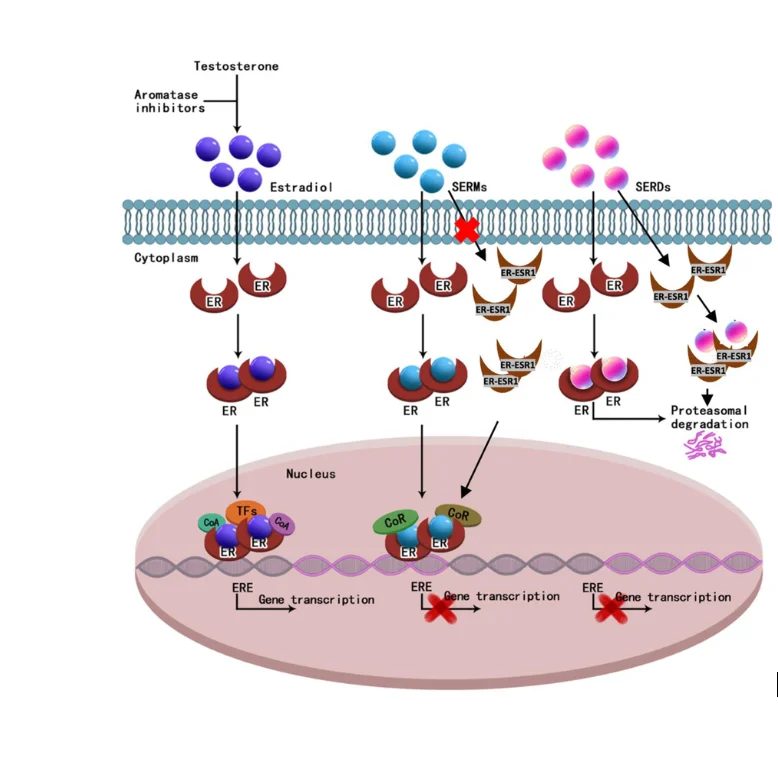
A 56% reduction in progression risk is unusually large for a hormone-receptor-positive breast cancer trial, and the patient-reported outcome data showing quality-of-life deterioration delayed by 14 months strengthens the case. FDA approves with a boxed or prominent warning on QT prolongation, restrictions on ribociclib co-administration, and a post-marketing commitment to deliver mature overall survival data by 2028. Guardant360 CDx is approved as companion diagnostic.