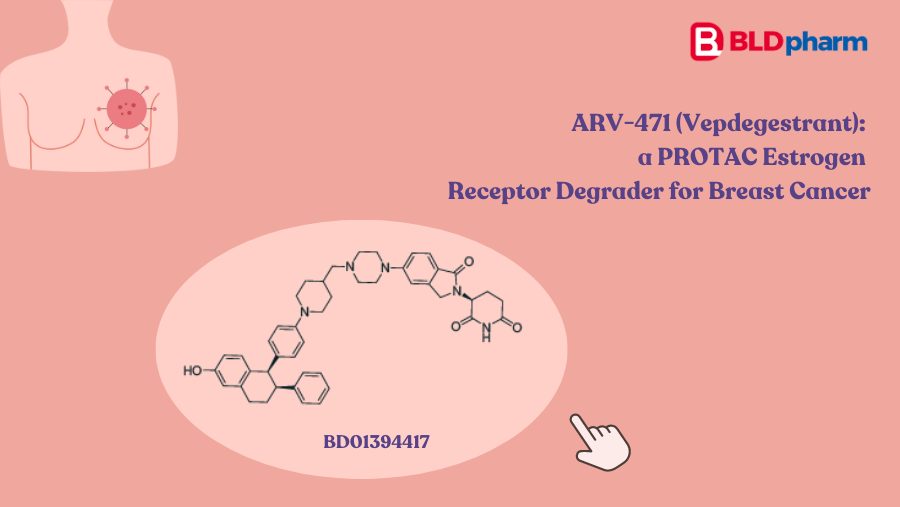
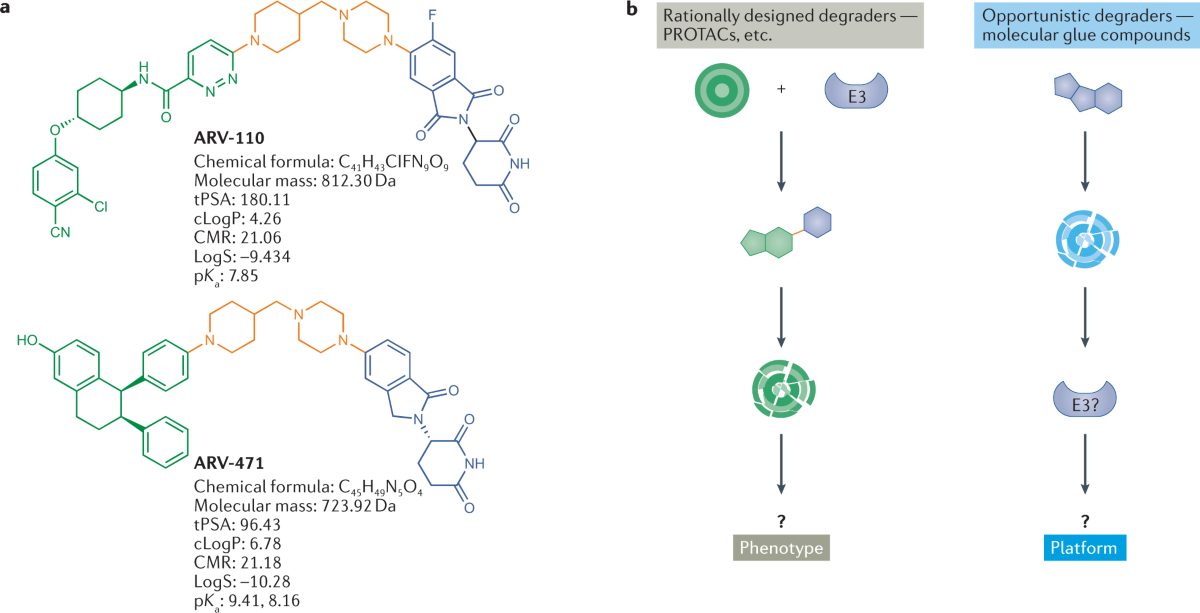
For 25 years, drug companies have tried to design medicines that don't just block harmful proteins but destroy them outright. On May 1, the U.S. Food and Drug Administration (FDA) approved the first one. VEPPANU, developed by biotech Arvinas and Pfizer, treats a hard-to-treat form of metastatic breast cancer by hijacking the cell's own garbage-disposal system to shred a mutated estrogen receptor that fuels the tumor.
The approval validates a class of drugs called PROTACs (proteolysis targeting chimeras), an idea first published in 2001 that had never produced a marketed medicine. It opens a regulatory path for similar drugs aimed at proteins that conventional medicines cannot touch, including drivers of cancer drug resistance, neurodegeneration, and inflammation. Roughly two dozen PROTACs from Arvinas, Kymera Therapeutics, Bristol Myers Squibb, Nurix, C4 Therapeutics, and Foghorn are now in human trials, and what happens to VEPPANU's launch will shape which of them get funded next.