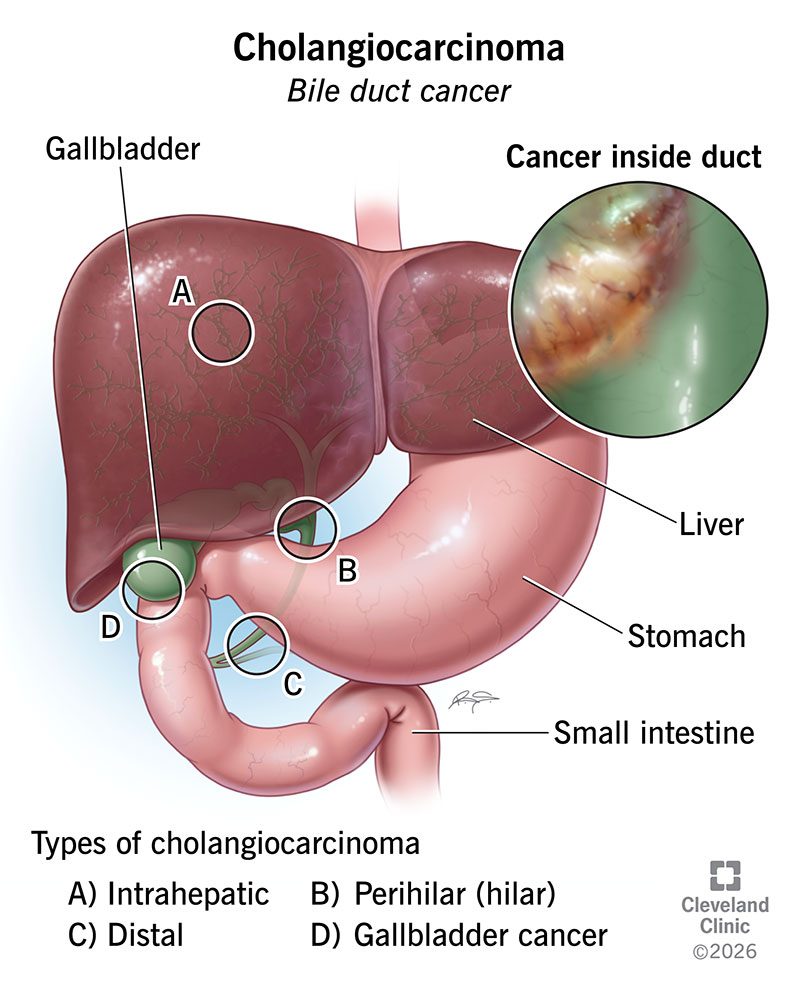
Patients with advanced bile duct cancer driven by a rare genetic fusion called NRG1 had nothing approved for them after chemotherapy failed. On May 8, the U.S. Food and Drug Administration (FDA) approved Bizengri (zenocutuzumab-zbco), made by Partner Therapeutics, as the first drug aimed at this small group. It works by blocking the abnormal protein the NRG1 fusion produces, which fuels tumor growth.
The approval is the seventh under the FDA Commissioner's National Priority Voucher (CNPV) pilot, a new pathway that compresses regulatory review for drugs the agency deems high priority. The CNPV review for Bizengri ran more than five months shorter than the standard goal date. Bile duct cancer, or cholangiocarcinoma, kills most patients within a year of an advanced diagnosis, and only a tiny share carry the NRG1 fusion.