For the roughly 30,000 Americans living with primary sclerosing cholangitis (PSC), the U.S. Food and Drug Administration has never approved a single drug. On May 4, 2026, that drought moved closer to ending: Mirum Pharmaceuticals reported that its experimental pill volixibat hit the primary endpoint of its Phase 2b VISTAS trial, cutting the relentless itch that defines the disease by a wide margin over placebo.
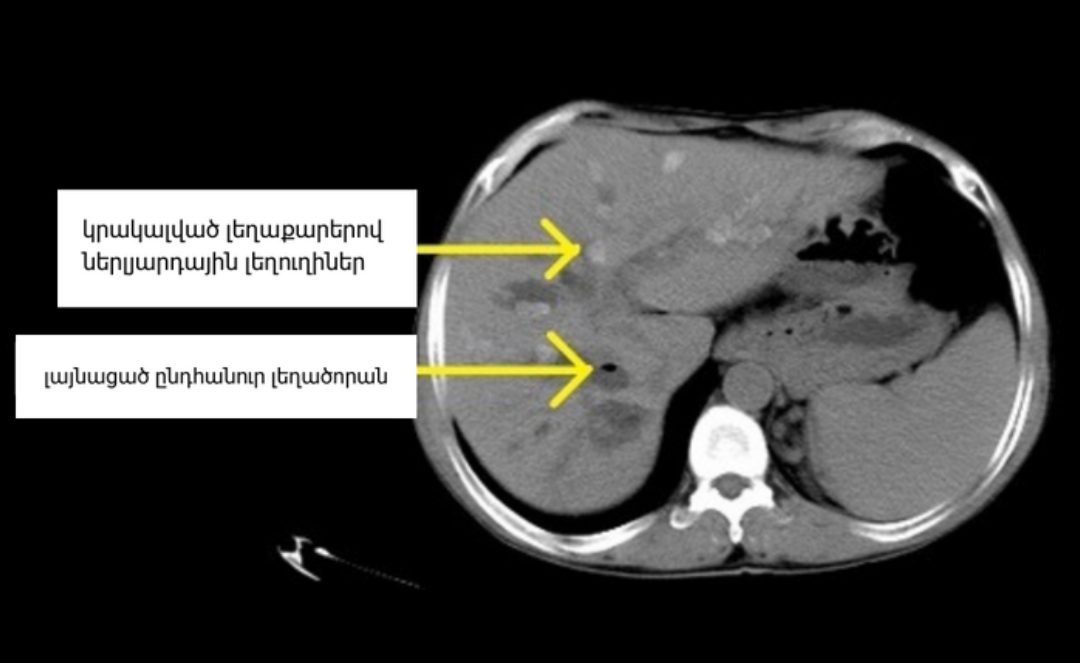
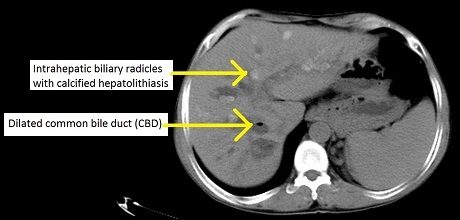
PSC is a chronic disease that scars the bile ducts inside and outside the liver, eventually pushing many patients toward liver transplant. Pruritus — a deep, intractable itch caused by bile acids backing up into the bloodstream — is one of its most disabling symptoms. With the VISTAS data in hand, Mirum says it will meet with the FDA this summer and file a New Drug Application (NDA) in the second half of 2026, putting the first-ever PSC therapy within reach of approval as soon as 2027.