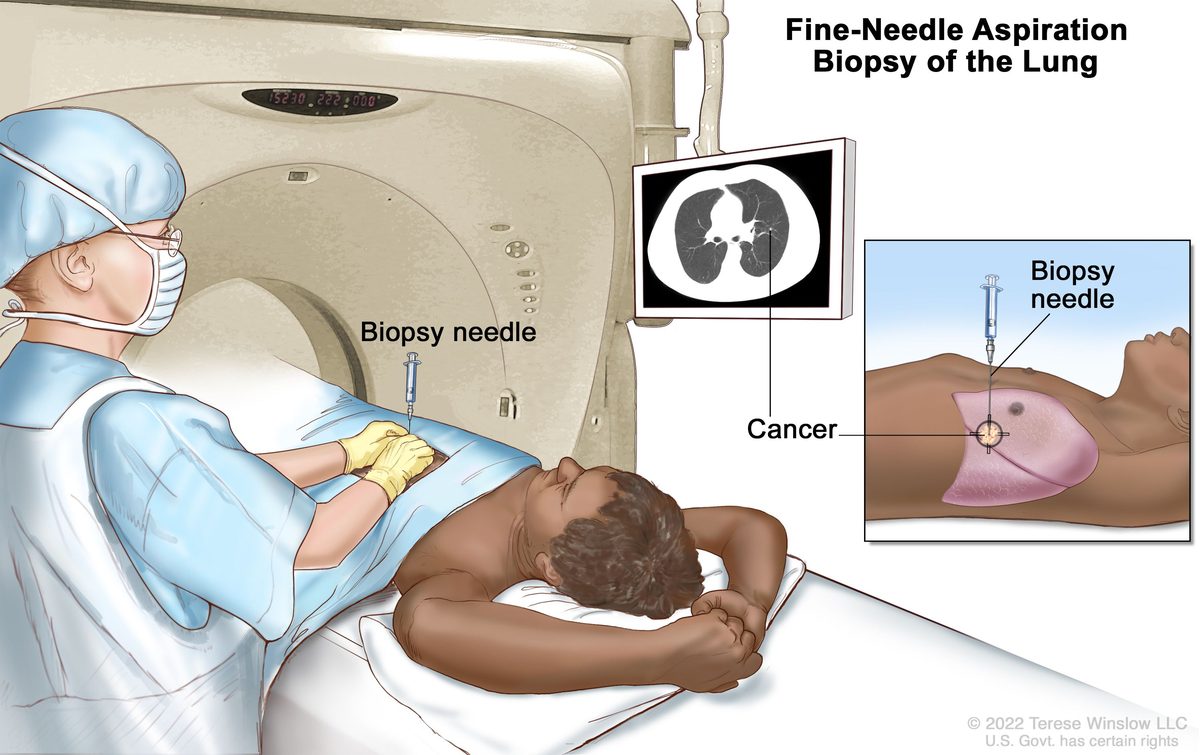
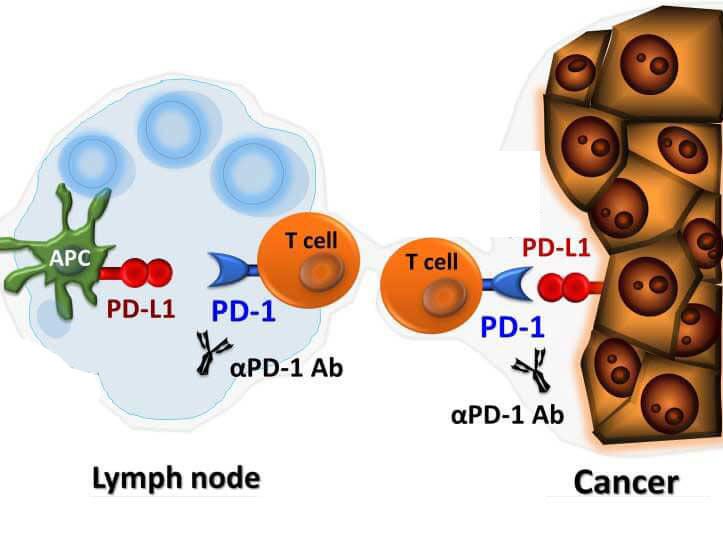
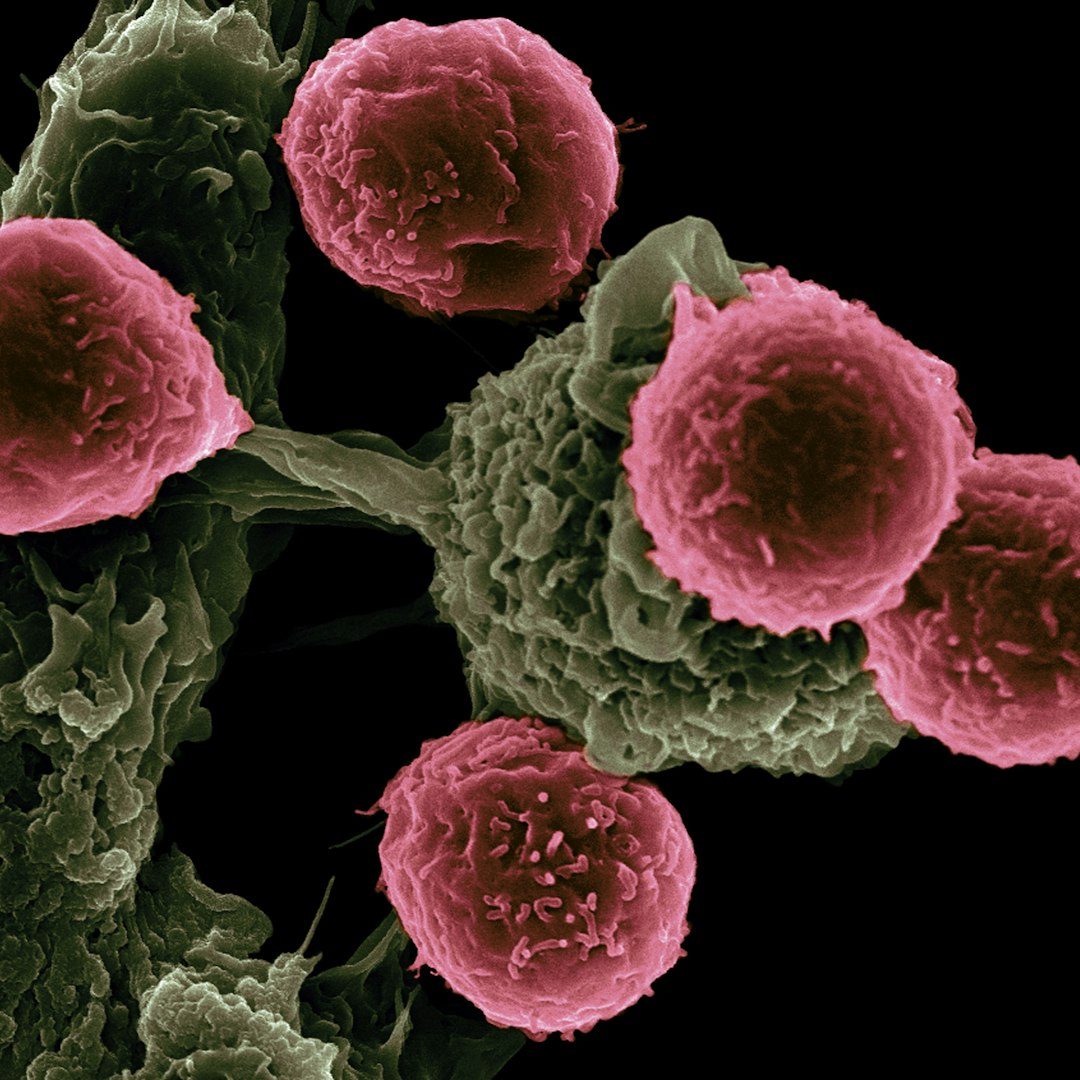
Tarlatamab becomes standard of care worldwide and moves into first-line treatment
With US traditional approval secured, China approved, and EU approval expected by mid-2026, tarlatamab achieves broad global availability. Three ongoing Phase III trials testing tarlatamab in earlier treatment lines — including as a first-line therapy combined with chemotherapy and immunotherapy — report positive results, expanding the eligible patient population several-fold. Analysts project global sales exceeding $2 billion annually. This is the path Amgen is actively building toward.