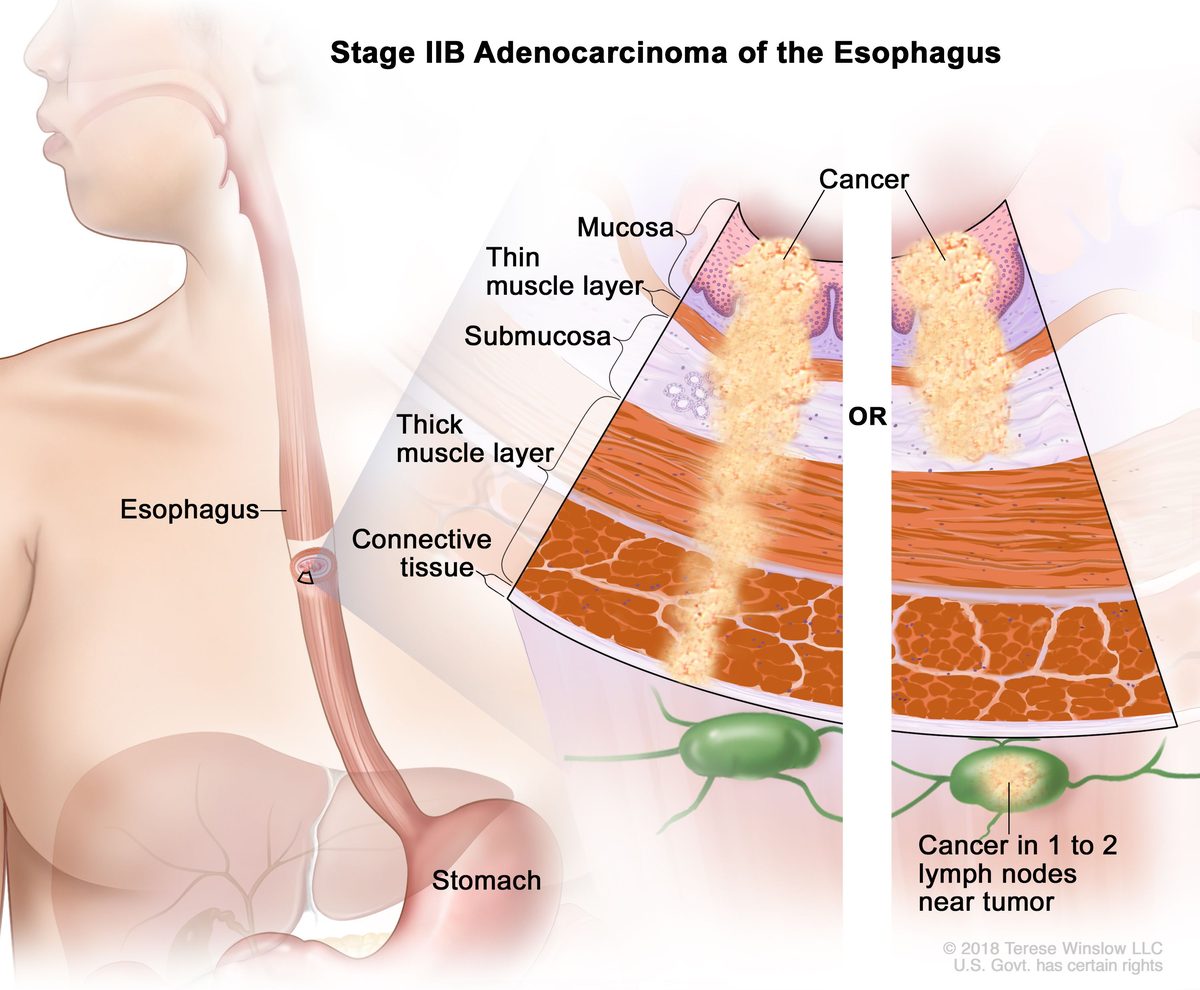
HS-20093 wins NMPA approval for esophageal cancer in 2027
Hansoh runs a confirmatory phase 3 trial against best supportive care or investigator's choice chemotherapy. With breakthrough status, NMPA accepts rolling submission and clears the drug for late-line use within 12 to 18 months. This is the base case if response rates from the registrational study hold above 30%.