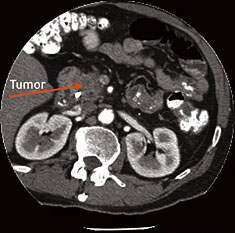
AI-PACED confirms benefit, REDMOD becomes standard for high-risk patients
If the prospective trial shows REDMOD-guided screening leads to more curable surgeries without flooding the system with false positives, hospitals adopt it for patients with new-onset diabetes, family history, or pancreatic cysts. Insurance coverage follows for the high-risk subgroup. Survival rates begin moving for the first time in a decade.