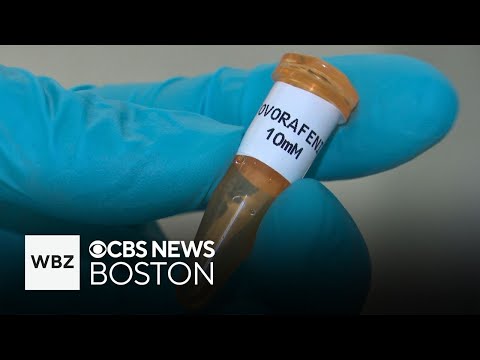
Servier closes the deal and accelerates Ojemda's expansion into front-line treatment
Servier completes the acquisition in the second quarter of 2026, as planned. With Servier's global infrastructure, the FIREFLY-2 Phase 3 trial — testing Ojemda as a first-line treatment rather than only for children whose tumors have already returned — completes enrollment and eventually yields positive results. This would significantly expand the eligible patient population and could push Ojemda well past its current $225-250 million revenue guidance. The existing licensing deal with Ipsen for markets outside the United States remains in place, creating a two-company global commercial footprint for the drug.