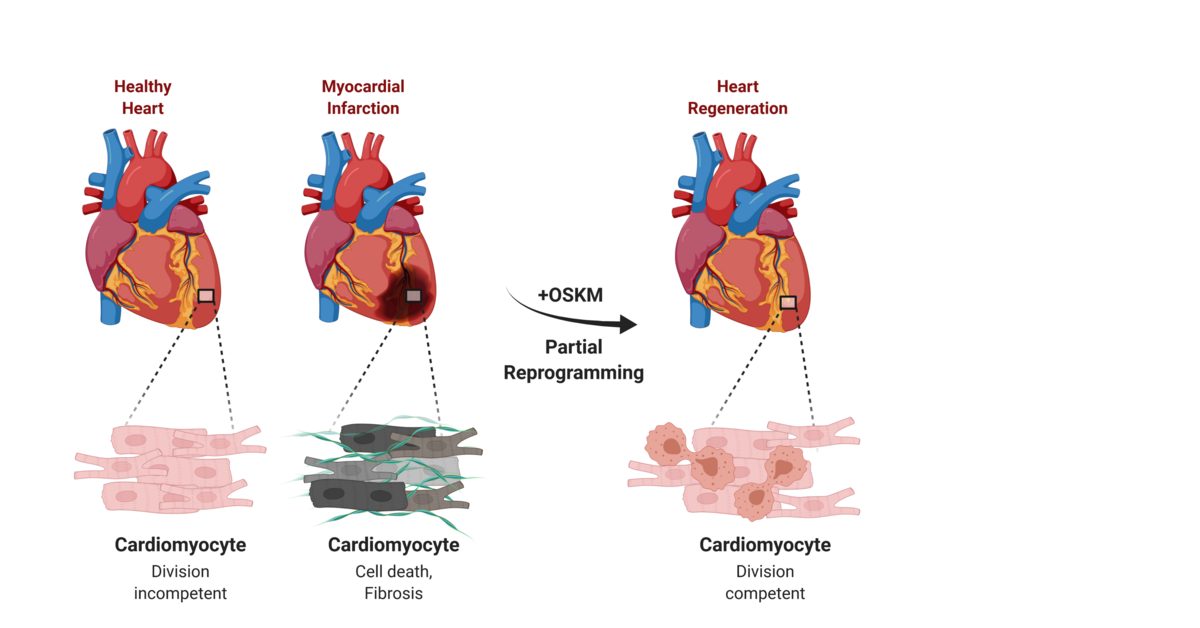
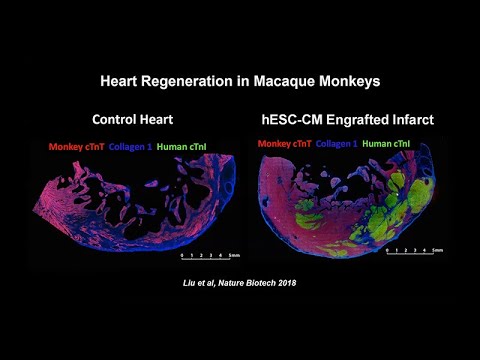
Stem Cell Patches Reach Clinical Practice by 2030
Mayo's minimally invasive patch delivery system completes trials successfully and wins FDA approval within four years. Hospitals begin offering the procedure to severe heart failure patients whose only current option is transplant. Insurance coverage follows FDA approval. The $1.9 billion cardiology stem cell market accelerates to $2.7 billion by 2029 as multiple institutions scale manufacturing of patient-specific tissue patches. Initial treatments cost $100,000+ but decline as techniques standardize. Cardiologists can identify ideal candidates: patients with advanced left ventricular enlargement who show high inflammation markers. Success depends on solving immunogenicity challenges and proving long-term integration of engineered tissue.