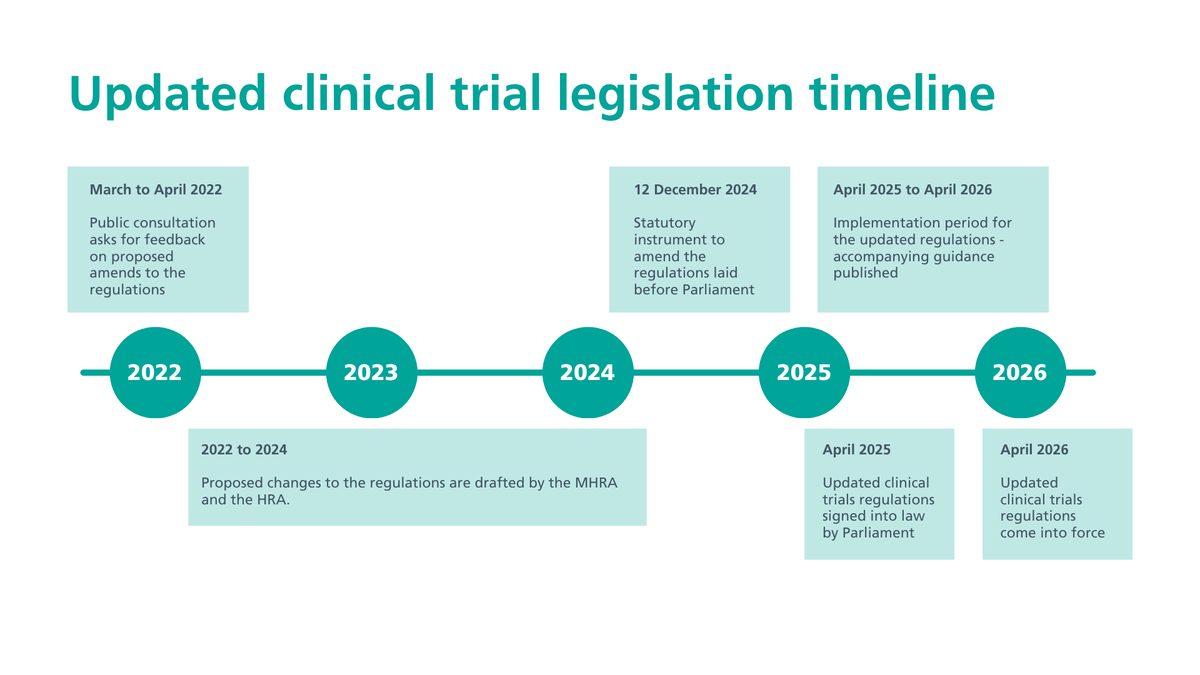
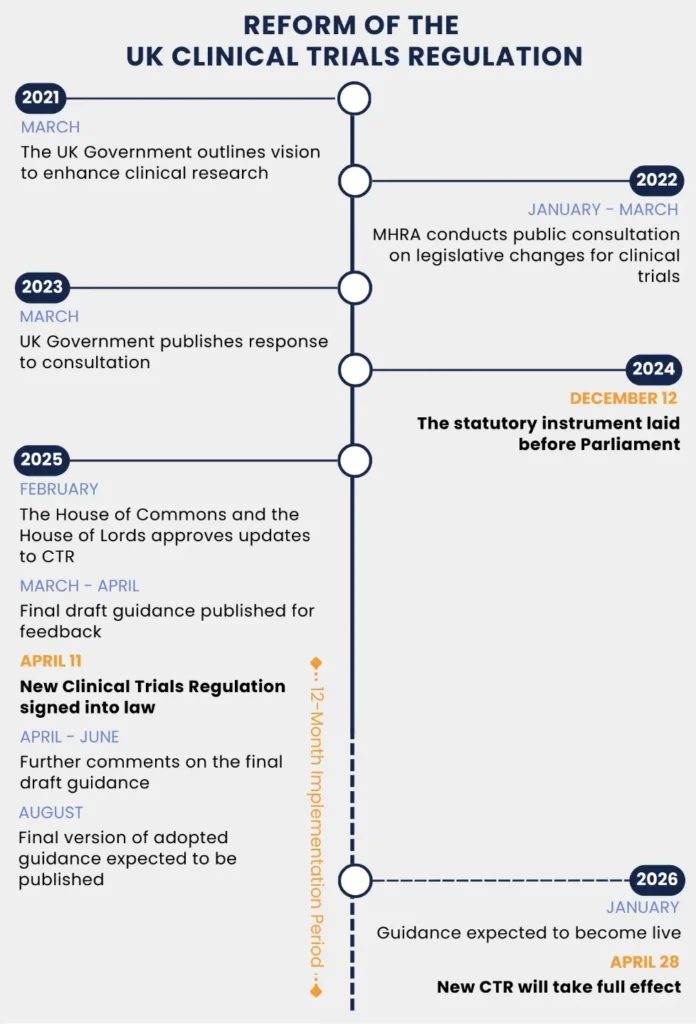
For more than two decades, running a clinical trial in the United Kingdom meant navigating a framework written in 2004 for a different era of medicine. On 28 April 2026, that framework was retired. New regulations from the Medicines and Healthcare products Regulatory Agency (MHRA) and Health Research Authority (HRA) introduce a fast-track route for lower-risk studies, formalise a seven-day approval pathway for trial modifications, and make registration and publication of summary results a legal requirement for the first time.
The shift matters because the UK had been losing ground as a trial destination. Combined safety and ethics review timelines have already fallen from 169 days to 122 days under preparatory work, and pharmaceutical sponsors who design global trials—including those run jointly with United States investigators—are now choosing where to place studies under a different rulebook. Faster, cheaper UK trials mean earlier patient access to experimental treatments; mandatory transparency means negative results no longer disappear.