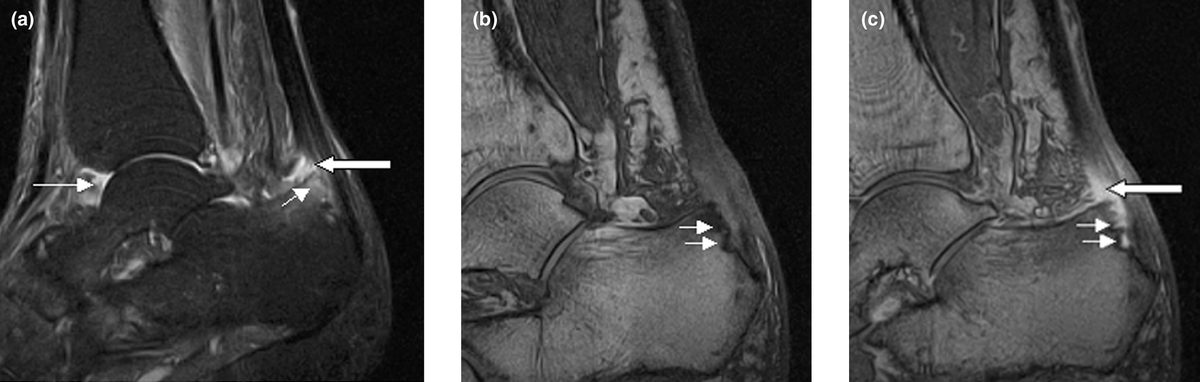
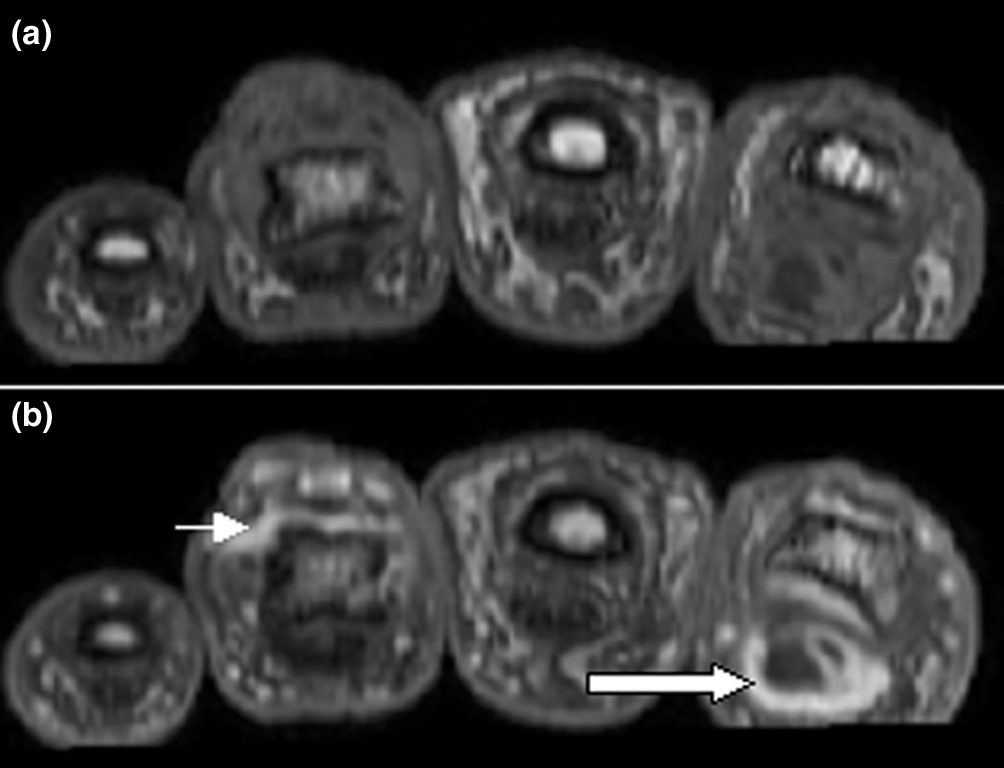
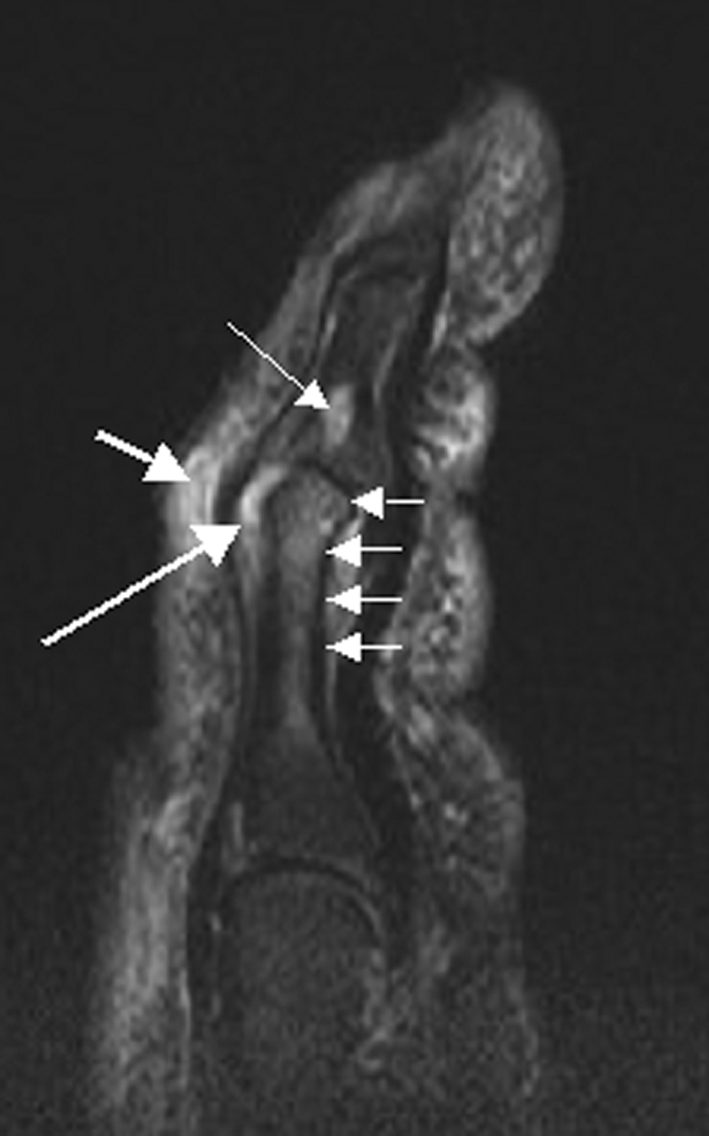
For most of the past decade, people with psoriatic arthritis who wanted a pill instead of an injection had one real option: Janus kinase (JAK) inhibitors, a class of oral drugs that the Food and Drug Administration (FDA) slapped with its strongest safety warning in 2021 after a major trial linked them to higher rates of heart attacks, blood clots, cancer, and death. On March 6, the FDA reached its decision deadline on whether to approve the first oral drug from a different class entirely—Bristol Myers Squibb's Sotyktu (deucravacitinib), a selective tyrosine kinase 2 (TYK2) inhibitor—for adults with active psoriatic arthritis, a condition that causes painful joint inflammation in roughly 1.5 million Americans.
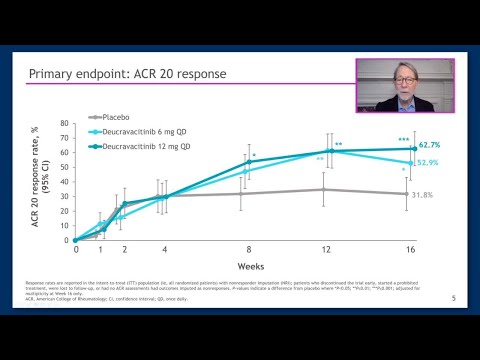
The distinction matters. Deucravacitinib works by binding to a different part of the enzyme than JAK inhibitors do, achieving selectivity that avoids the off-target effects that triggered the FDA's 2021 warnings. In two large trials, about 54% of patients on the drug saw at least 20% improvement in joint symptoms at 16 weeks, compared with 34–39% on placebo, with responses holding through a full year. If approved, Sotyktu would give rheumatologists and patients the first oral option in a new drug class for psoriatic arthritis—and give Bristol Myers Squibb a critical revenue expansion for a drug it projects could eventually generate $4 billion in annual sales.