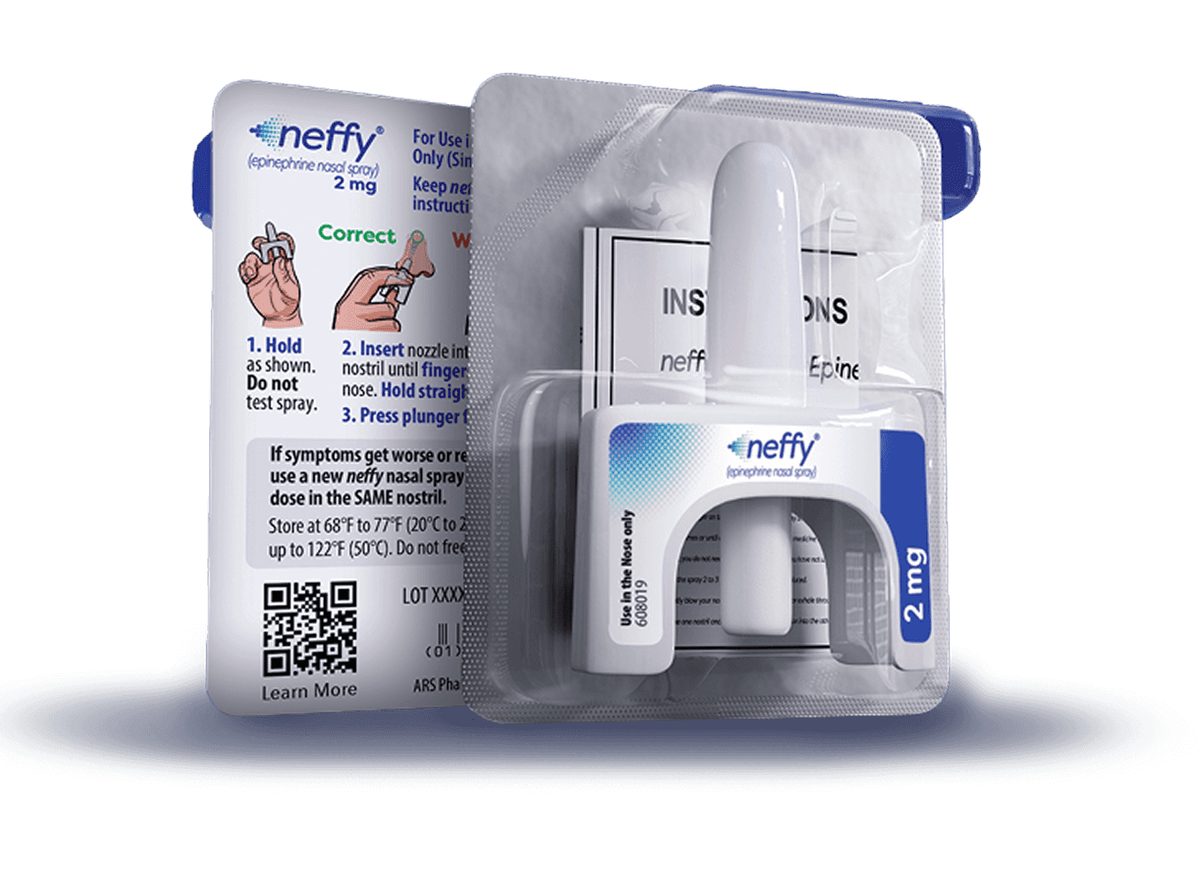
For nearly four decades, Americans facing life-threatening allergic reactions had exactly one option: stab themselves with a needle. The EpiPen and its competitors delivered epinephrine effectively, but needle phobia kept roughly 30% of patients from using their prescribed devices during emergencies—contributing to preventable deaths. Now the needle era is ending. The Food and Drug Administration (FDA) approved the first needle-free alternative, a nasal spray called neffy, in August 2024. On February 2, 2026, the FDA issued a Complete Response Letter for Anaphylm, a dissolvable film placed under the tongue, rejecting the application but limiting deficiencies to human factors and packaging rather than safety or efficacy concerns. Aquestive Therapeutics expects to resubmit in the third quarter of 2026 after conducting a redesigned human factors validation study and a supporting pharmacokinetics study.
The stakes extend beyond convenience. An estimated 33 million Americans carry severe allergy risks, with food allergies alone triggering 30,000 emergency room visits and 150 to 200 deaths annually. Delayed epinephrine use is a leading risk factor in fatal reactions. Meanwhile, neffy is expanding internationally—the European Commission approved EURneffy in August 2024, and in February 2026, the European Medicines Agency's Committee for Medicinal Products for Human Use issued a positive opinion for neffy in children. If Anaphylm gains approval following its resubmission and both needle-free options gain market traction, the treatment gap created by fear of needles could narrow substantially—potentially saving lives that current delivery methods cannot reach.