FDA Approves, Launch Begins in Q1 2026
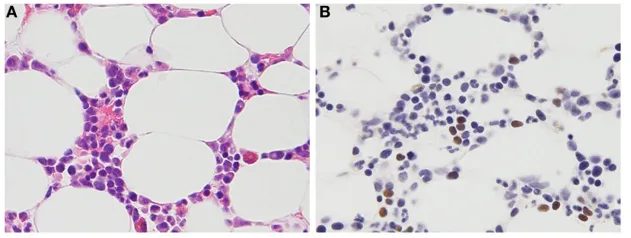
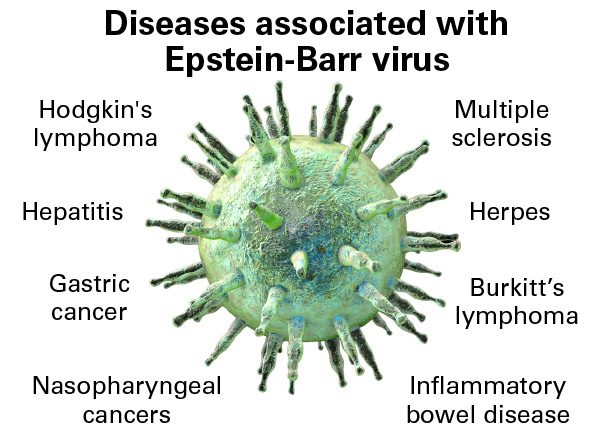
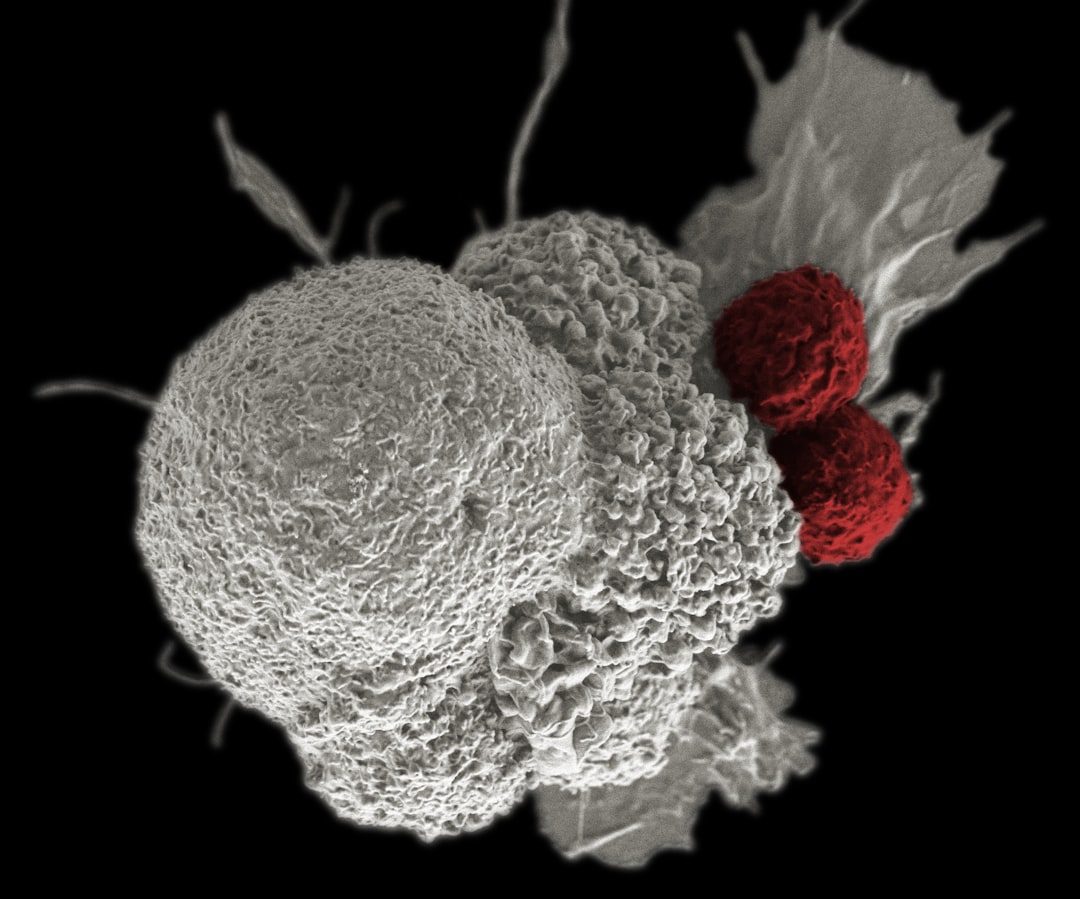
The FDA approves tabelecleucel, making it the first therapy for EBV+ PTLD after prior treatment. Pierre Fabre triggers a $40 million milestone payment to Atara and begins U.S. commercialization, building on European experience since 2022. The approval validates the off-the-shelf allogeneic T-cell therapy model, potentially opening pathways for other companies developing similar platforms. Given that the agency already confirmed no clinical or safety issues and manufacturing problems have been resolved, approval appears likely.