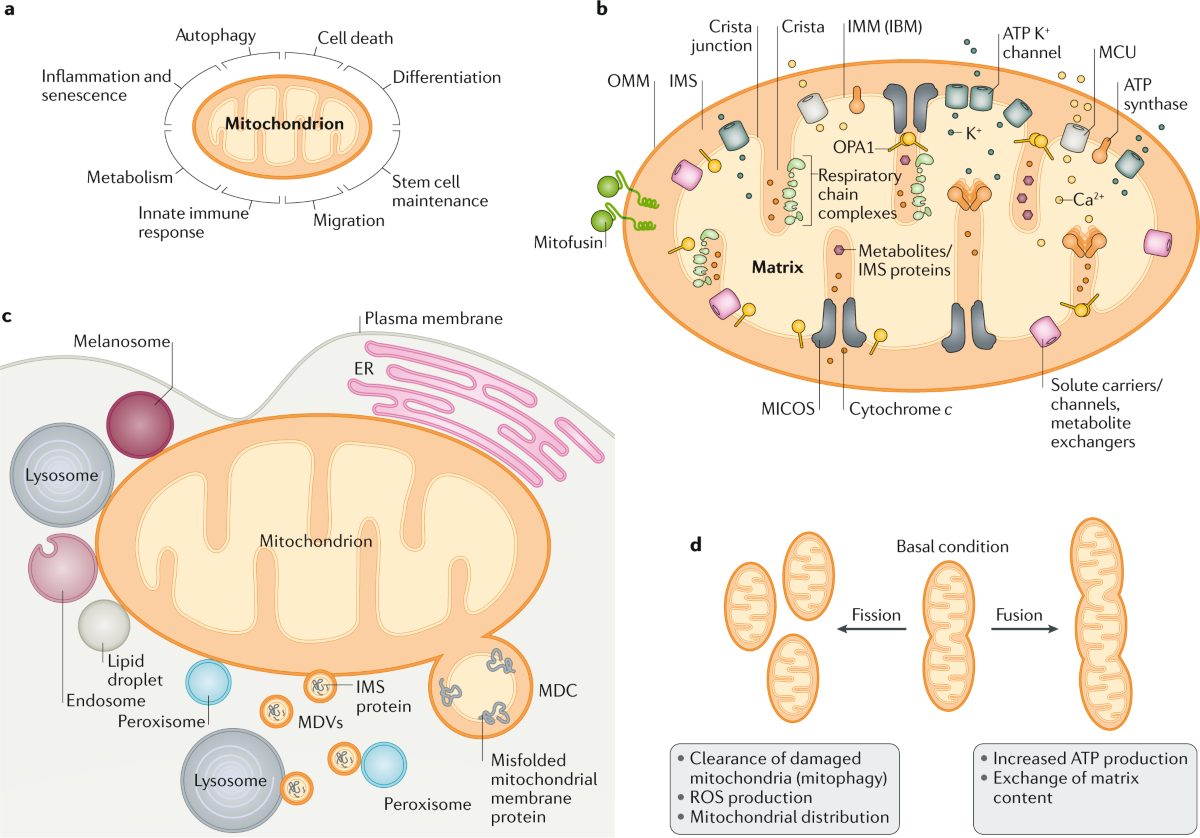
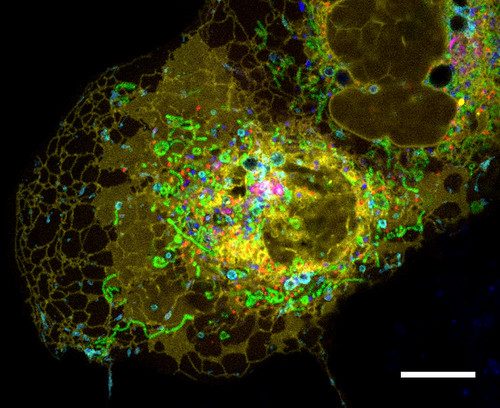
For most of modern medicine's history, diseases caused by defective mitochondria — the structures inside cells that generate energy — have been essentially untreatable. Doctors could manage symptoms, but the broken power plants themselves were beyond reach. Now, within a six-month span, the United States Food and Drug Administration (FDA) has approved the first two drugs targeting mitochondrial diseases, and a Chinese research team has demonstrated a method to physically transplant healthy mitochondria into diseased cells with sixteen times the efficiency of previous approaches.
The transplantation technique, published in Cell on March 18, 2026, wraps donor mitochondria in membranes harvested from red blood cells — a disguise that tricks recipient cells into accepting the foreign organelles rather than destroying them. In mice with Leigh syndrome, a fatal childhood neurological disease, the therapy extended survival by roughly 20%. In a Parkinson's disease model, treated mice recovered near-normal motor function. The results sit alongside two recent FDA approvals — elamipretide for Barth syndrome in September 2025 and doxecitine-doxribtimine for thymidine kinase 2 deficiency in November 2025 — marking a turning point for a field that had zero approved therapies as recently as mid-2025.