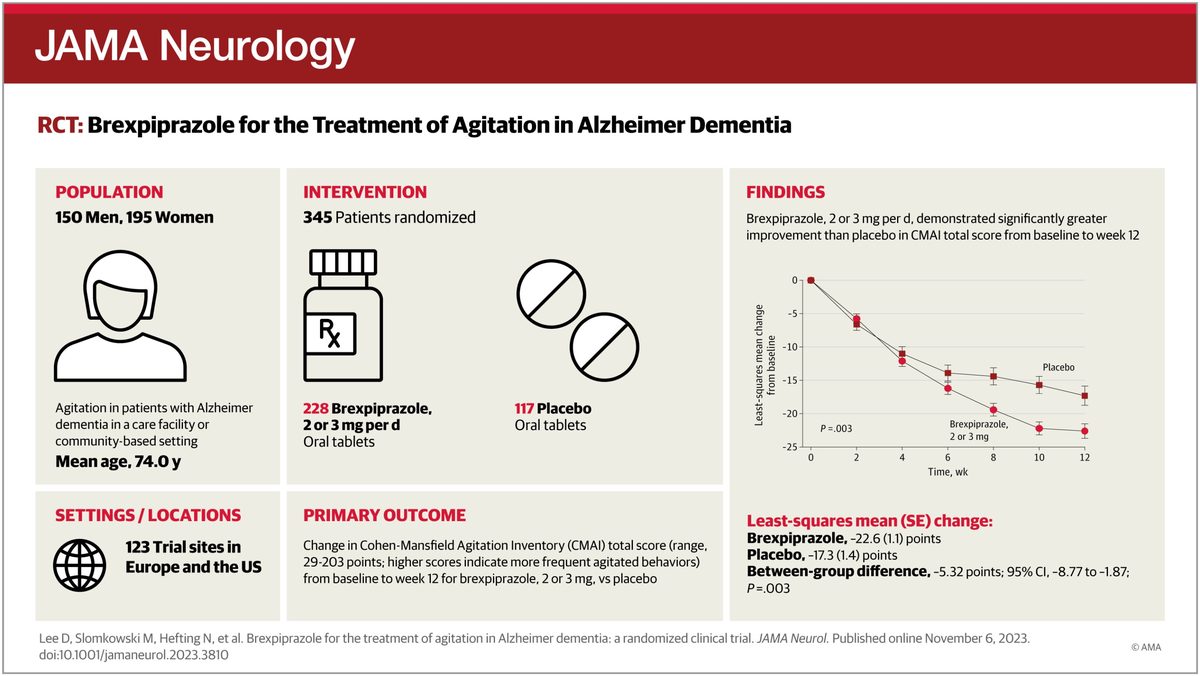
Until May 2023, no drug was approved in the United States to treat the agitation that affects up to three-quarters of people with Alzheimer's disease. Doctors reached for antipsychotics off-label, a class of medicines that carries a black-box warning for increased death risk in older dementia patients. On April 30, 2026, the Food and Drug Administration (FDA) approved Axsome Therapeutics' Auvelity for the indication, doubling the menu of approved options from one to two. In its own press announcement, the FDA described the action as approving the 'first non-antipsychotic drug to treat agitation associated with dementia' — a framing that underscores what distinguishes Auvelity from the incumbent treatment, Rexulti.
Auvelity is an oral pill combining dextromethorphan, the cough-suppressant ingredient, with bupropion, an older antidepressant that slows dextromethorphan's breakdown so it can act on the brain. In two pivotal trials, patients on Auvelity relapsed into agitation at 8.4 percent versus 28.6 percent on placebo. Axsome moved quickly to commercial launch: the company had already doubled its salesforce to 600 representatives in the months before approval and activated its Auvelity OnMySide patient support program — offering savings cards for commercially insured patients, drug samples, and prior authorization assistance — on the day of approval. AXSM shares surged on the news. The company is scheduled to report Q1 2026 financial results on May 4, which will be the first quarterly disclosure following the new indication.